A Comprehensive Look at the Benefits of Lighting for Reptiles
Image sourced from gaurani.almightywind.info
Introduction
The importance of light in animal care is often undervalued, light plays an important role in the health of all life on earth, and captive reptiles are no exception. As we discuss light, it is important to know exactly what it is so we may understand what value it has for our animals.
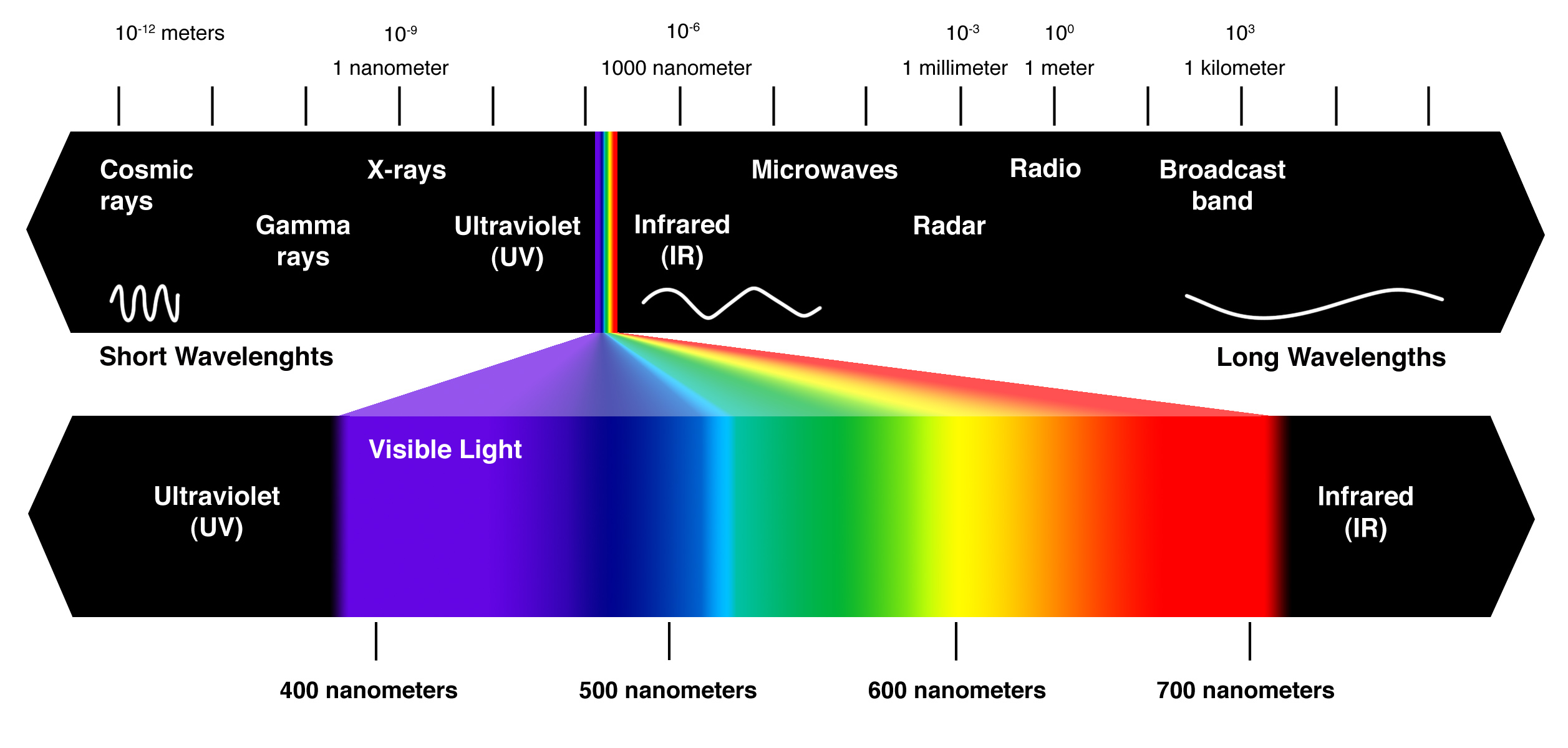
The spectrum of light is composed of electromagnetic waves, these waves can be divided into several ranges or bands, and is measured in nanometres (nm). Ultraviolet radiation extends from 100 to 400 nm and is subdivided into three distinct bands; UV-C (lowest band: 100-280 nm), UV-B (middle band: 280-315 nm), and UV-A (upper band: 315-400 nm). Visible radiation (400-700 nm) is what we consider visible light, different frequencies of light in this range are interpreted as different colours. Above visible radiation is Infrared (700- 3,200 nm) which we interpret as heat.
The spectrum of light is composed of electromagnetic waves, these waves can be divided into several ranges or bands, and is measured in nanometres (nm). Ultraviolet radiation extends from 100 to 400 nm and is subdivided into three distinct bands; UV-C (lowest band: 100-280 nm), UV-B (middle band: 280-315 nm), and UV-A (upper band: 315-400 nm). Visible radiation (400-700 nm) is what we consider visible light, different frequencies of light in this range are interpreted as different colours. Above visible radiation is Infrared (700- 3,200 nm) which we interpret as heat.
Image sourced from nbertoa.wordpress.com
The natural sunlight that reaches the earth's surface includes near infrared, visible light, UV-A and some UV-B, whereas wavelengths below 285 nm and above 2,500 nm are normally filtered by the earth's atmosphere, protecting us from dangerous radiation.
Essentially being solar-powered animals, light means something different to reptiles than it does to humans. As ectothermic (‘cold blooded’) animals, reptiles maintain their optimal body temperatures behaviourally, and as such depend directly on the infrared (IR) portion of the light spectrum as a source of heat (via basking – known as heliothermy) and indirectly on convection (from warm substrates and warm or cool burrows – known as thigmothermy). Warming light is a pivotal factor in regulating reptile microclimates and microhabitats and is a critical underpinning for the wellbeing of reptiles. Not only is the IR portion of solar energy important, but so is the visible and ultraviolet (UV) spectrum, as well as other characteristics of light such as intensity, colour, frequency and duration (light cycles). This shall be discussed in depth below.
Circadian Rhythm
Many reptile keepers are aware of the importance of providing a day-night light cycle, but few are aware of the full extent as to the impact that light cycles have on animals, as well as the effect that the different forms of light can have.
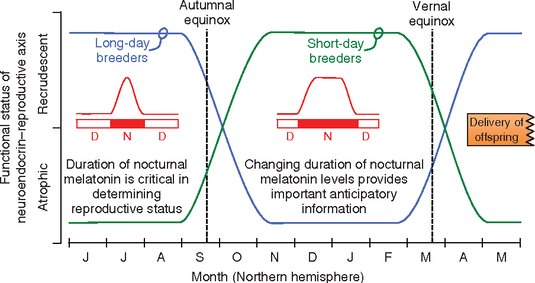
Melatonin is the hormone that is predominantly responsible for keeping the body’s inherent circadian rhythms in synchronicity with light/dark cycles. Melatonin is a hormone of darkness and its secretion increases up to 10 fold during the darkness of night, then falls back to low levels during the day. One example to demonstrate the importance of light cycles can be seen in animals that breed seasonally. ‘Short day’ breeders rely on steadily increasing concentrations of melatonin while ‘long day’ breeders rely on the progressively decreasing concentrations of melatonin to trigger reproduction. Therefore, increasing/decreasing day lengths are important considerations for seasonally correct reproductive cycles of many animals, including reptiles.
Melatonin is the hormone that is predominantly responsible for keeping the body’s inherent circadian rhythms in synchronicity with light/dark cycles. Melatonin is a hormone of darkness and its secretion increases up to 10 fold during the darkness of night, then falls back to low levels during the day. One example to demonstrate the importance of light cycles can be seen in animals that breed seasonally. ‘Short day’ breeders rely on steadily increasing concentrations of melatonin while ‘long day’ breeders rely on the progressively decreasing concentrations of melatonin to trigger reproduction. Therefore, increasing/decreasing day lengths are important considerations for seasonally correct reproductive cycles of many animals, including reptiles.
Image by Reiter, et al., (2010)
Although all animals possess biological clocks, these clocks must be reset daily as the natural pacemaker is either slightly faster or slower than a 24-hour period, depending on species and life-stage. When circadian rhythms are left to free-run in either constant light or constant darkness, they fall out of sync with natural cycles, this can cause disruption to sleep, mood, behaviour and various biological and physiological processes. Therefore, these rhythms must be repeatedly reset to synchronise with nature’s cycle.
Maintaining this synchronisation is known as “entrainment”. Light exposure to specific wavelengths is the strongest environmental cue (known as a zeitgeber) for the entrainment of circadian rhythms, and light intensity is also important for the regulation of free-running periods in many species of animals, but it should be made aware that ambient temperature cycle is also an effective zeitgeber for entrainment of circadian rhythms, so the inclusion of ambient temperature cycles should be considered alongside light cycles.
Light has many different characteristics (e.g., wavelength, hue, intensity, duration of the exposure, time of day), which can all impact on the animal. For instance, blue light elicits the strongest effects on circadian rhythms. Cryptochromes are photoreceptors present in plants and animals, they are involved in circadian rhythms and specifically react to blue light. In the wild they are activated at dawn, causing the suppression of melatonin, and preventing its secretion by the pineal gland. Exposure to blue light can affect many physiologic functions, highlighting the importance of providing correct light at appropriate times of day. Blue light is particularly problematic as it is most damaging to the retina. Blue light (especially in the 480 nm spectrum:- common in LED's) can bleach the visual pigments found in rod cells (a type of photoreceptor), leading to loss of vision. This can occur with either very bright blue light over short-term exposure, or with lower levels of blue light over a long-term exposure.
Even blind animals are impacted by the many different characteristics of light. This is because the retina houses light sensitive cells separate to those responsible for vision, these are known as melanopsin-expressing retinal ganglion cells, and play no role in visual perception, they only inform the pineal gland as to the presence and intensity of light, thereby modulating melatonin production. The presence of these melanopsin cells allow for a relatively normal lifestyle in blind individuals, in spite of the lack of normal visual input. However, issues can occur with those that lack a retina or lack eyes, causing the circadian system to free-run.
In reptiles, the pineal gland is also directly light-sensitive. All reptiles except crocodilians have pineal organs that lie on the brain but do not exit the skull. Some lizards (e.g. iguanians) have pineal organs that pass through the skull and form the parietal eye. The pineal gland is a central component in the regulation of circadian rhythmicity of reptiles, while the parietal eye is present only in some lizard species and in the tuatara (Sphenodon punctatus). The parietal eye has its own primitive lens, cornea, and retina and is often called a “third eye”, although it is not used for vision.
Maintaining this synchronisation is known as “entrainment”. Light exposure to specific wavelengths is the strongest environmental cue (known as a zeitgeber) for the entrainment of circadian rhythms, and light intensity is also important for the regulation of free-running periods in many species of animals, but it should be made aware that ambient temperature cycle is also an effective zeitgeber for entrainment of circadian rhythms, so the inclusion of ambient temperature cycles should be considered alongside light cycles.
Light has many different characteristics (e.g., wavelength, hue, intensity, duration of the exposure, time of day), which can all impact on the animal. For instance, blue light elicits the strongest effects on circadian rhythms. Cryptochromes are photoreceptors present in plants and animals, they are involved in circadian rhythms and specifically react to blue light. In the wild they are activated at dawn, causing the suppression of melatonin, and preventing its secretion by the pineal gland. Exposure to blue light can affect many physiologic functions, highlighting the importance of providing correct light at appropriate times of day. Blue light is particularly problematic as it is most damaging to the retina. Blue light (especially in the 480 nm spectrum:- common in LED's) can bleach the visual pigments found in rod cells (a type of photoreceptor), leading to loss of vision. This can occur with either very bright blue light over short-term exposure, or with lower levels of blue light over a long-term exposure.
Even blind animals are impacted by the many different characteristics of light. This is because the retina houses light sensitive cells separate to those responsible for vision, these are known as melanopsin-expressing retinal ganglion cells, and play no role in visual perception, they only inform the pineal gland as to the presence and intensity of light, thereby modulating melatonin production. The presence of these melanopsin cells allow for a relatively normal lifestyle in blind individuals, in spite of the lack of normal visual input. However, issues can occur with those that lack a retina or lack eyes, causing the circadian system to free-run.
In reptiles, the pineal gland is also directly light-sensitive. All reptiles except crocodilians have pineal organs that lie on the brain but do not exit the skull. Some lizards (e.g. iguanians) have pineal organs that pass through the skull and form the parietal eye. The pineal gland is a central component in the regulation of circadian rhythmicity of reptiles, while the parietal eye is present only in some lizard species and in the tuatara (Sphenodon punctatus). The parietal eye has its own primitive lens, cornea, and retina and is often called a “third eye”, although it is not used for vision.
Image originally sourced from burrard-lucas.com
Light and Reptilian Immunity
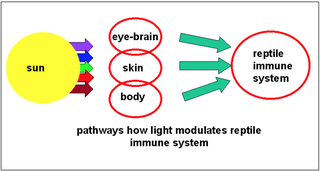
One particularly important consideration of appropriate light provision for reptiles is that light is an important factor to maintaining a well-regulated immune system. It needs to be emphasised that improving reptilian lighting environments is a critical factor in the creation of appropriate habitats for captive reptiles. Circadian rhythms and the production of the two neuro-hormones; melatonin and serotonin, directly modulate the reptilian immune system. Light also serves to dry the skin, providing a less comfortable surface for the growth of bacterial pathogens while promoting the growth of defensive bacterial flora. Finally, UV light itself has a direct disinfectant action, destroying pathogens and helping to maintain hygiene standards. Additionally, encouraging movement through enrichment, such as the provision of a proper basking area also encourages body movements and gut peristalsis to prevent bacterial overgrowth.
Immune cells under the skin react directly to the deep-penetrating (red) portion of the light spectrum and modifies immune function. Further, UV-B regulates vitamin D3 synthesis in the skin (the primary source of D3 in many reptiles). This nutrient can help stimulate the production of antibacterial agents. D3 also acts as an anti-carcinogen, helping to counteract the effects of carcinogens and inhibiting the development of cancer. The direct role of D3 on the immune system is often overlooked, with focus falling squarely on its role in calcium regulation. Also noteworthy is the fact that calcium itself plays an important role in long-distance immune cell communication. Therefore, synthesis of optimal levels of vitamin D3 via UV-B is important for mineral metabolism and also for immune regulation.
Immune cells under the skin react directly to the deep-penetrating (red) portion of the light spectrum and modifies immune function. Further, UV-B regulates vitamin D3 synthesis in the skin (the primary source of D3 in many reptiles). This nutrient can help stimulate the production of antibacterial agents. D3 also acts as an anti-carcinogen, helping to counteract the effects of carcinogens and inhibiting the development of cancer. The direct role of D3 on the immune system is often overlooked, with focus falling squarely on its role in calcium regulation. Also noteworthy is the fact that calcium itself plays an important role in long-distance immune cell communication. Therefore, synthesis of optimal levels of vitamin D3 via UV-B is important for mineral metabolism and also for immune regulation.
Image by Brames, et al., (2007).
Reptile Vision
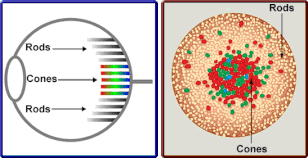
When considering lighting for reptiles, it should be acknowledged that reptiles perceive light differently than humans and other mammals. Vision is made possible through the existence of two types of photoreceptors in the eye:- rods and cones, which are distinguished from each-other by their shapes (from which their names are derived). These photoreceptors are found within the retina, located at the back of the eye and are responsible for converting light signals into electronic signals, allowing us to visually perceive the world around us.
Rods are sensitive to the detection of shapes and movement and are sensitive to light, so are responsible for vision in low light situations such as at night. However, rods have very low spatial acuity, meaning vision from these photoreceptors lacks sharpness. Furthermore, they do not allow for colour vision. Rods are in their greatest numbers around the 'periphery' of the retina, meaning they're most active in our peripheral vision; the corner of our vision. Conversely, cones work best in brighter conditions, as they are relatively insensitive to light. They have very high spatial acuity meaning vision is sharp and focused; colour vision is also made possible with the presence of cones. The central part of the eye (the fovea) is populated exclusively by cones, processing the information of what you are directly looking at as opposed to what's perceived in the corner of your eye.
Retinas of almost all vertebrates possess rods, but not all vertebrates have cones. Surprisingly, geckos, most of which are nocturnal, lack rods, but instead have very sensitive cones for night colour vision. Reptiles generally have very good colour vision, better so than humans, but comparatively few mammals see in colour, or only have limited colour vision, with the exception of humans and some other primates.
Generally speaking, mammals are typically dichromatic (possess only two types of cones; green and blue), except for Humans and Old World primates, who are trichromatic (three cone types; red, green, and blue). Human vision covers 400–700 nm on the electromagnetic spectrum (visible light), while reptiles are tetrachromatic animals, meaning that they possess 4 types of cones (sensitive to red, green, blue and UV-A), possessing an additional cone that allows reptiles to see into the UV-A spectra (below 400 nm). Additionally, the red, green, and blue cones of reptiles have shifted peak sensitivities when compared to those of humans, so they do not perceive colour in quite the same way we do. This can have implications with using artificial lighting optimised for human vision. Some species, especially night active and crepuscular species, lack red cones, but still have very good colour vision, even in very dim light that would render humans colour-blind.
Rods are sensitive to the detection of shapes and movement and are sensitive to light, so are responsible for vision in low light situations such as at night. However, rods have very low spatial acuity, meaning vision from these photoreceptors lacks sharpness. Furthermore, they do not allow for colour vision. Rods are in their greatest numbers around the 'periphery' of the retina, meaning they're most active in our peripheral vision; the corner of our vision. Conversely, cones work best in brighter conditions, as they are relatively insensitive to light. They have very high spatial acuity meaning vision is sharp and focused; colour vision is also made possible with the presence of cones. The central part of the eye (the fovea) is populated exclusively by cones, processing the information of what you are directly looking at as opposed to what's perceived in the corner of your eye.
Retinas of almost all vertebrates possess rods, but not all vertebrates have cones. Surprisingly, geckos, most of which are nocturnal, lack rods, but instead have very sensitive cones for night colour vision. Reptiles generally have very good colour vision, better so than humans, but comparatively few mammals see in colour, or only have limited colour vision, with the exception of humans and some other primates.
Generally speaking, mammals are typically dichromatic (possess only two types of cones; green and blue), except for Humans and Old World primates, who are trichromatic (three cone types; red, green, and blue). Human vision covers 400–700 nm on the electromagnetic spectrum (visible light), while reptiles are tetrachromatic animals, meaning that they possess 4 types of cones (sensitive to red, green, blue and UV-A), possessing an additional cone that allows reptiles to see into the UV-A spectra (below 400 nm). Additionally, the red, green, and blue cones of reptiles have shifted peak sensitivities when compared to those of humans, so they do not perceive colour in quite the same way we do. This can have implications with using artificial lighting optimised for human vision. Some species, especially night active and crepuscular species, lack red cones, but still have very good colour vision, even in very dim light that would render humans colour-blind.
An image to demonstrate what a human sees (left) compared to what a tetrachromatic animal, such as a bird or reptile sees (right).
Additionally, some reptiles can see precisely even at night by exploiting the IR portion of the spectrum. Some snake species (pit vipers, boid and pythonid snakes) use IR sensors located on their faces (pit organs) to detect the IR spectra of light (heat) and allow them to see clearly in total darkness.
Due to our differences in vision, it is an important consideration to distinguish the different lighting requirements of humans and reptiles. Artificial lighting has been optimised for human sensitivities rather than reptilian demands, so, for instance, although lack UV-A or other parts of the spectra do not bother humans, it may dramatically affect reptiles. As well as colour perception, reptiles may also see hues and illuminance in a way that we can hardly imagine. Behavioural studies show that UV spectra and reptile-correct colour rendering of artificial light settings is not only necessary for conspecific, interspecific, and intersexual recognition, but that brightness and contrast also are critical for motion perception and foraging, and likely also for maintaining a sense of wellbeing (“happiness”) in reptiles.
Due to our differences in vision, it is an important consideration to distinguish the different lighting requirements of humans and reptiles. Artificial lighting has been optimised for human sensitivities rather than reptilian demands, so, for instance, although lack UV-A or other parts of the spectra do not bother humans, it may dramatically affect reptiles. As well as colour perception, reptiles may also see hues and illuminance in a way that we can hardly imagine. Behavioural studies show that UV spectra and reptile-correct colour rendering of artificial light settings is not only necessary for conspecific, interspecific, and intersexual recognition, but that brightness and contrast also are critical for motion perception and foraging, and likely also for maintaining a sense of wellbeing (“happiness”) in reptiles.
An image by Brames, et al., (2007) to demonstrate what artificial lighting (non-continuous spectrum or lacking UV-A) may be doing to reptile vision. When only one of the colours we perceive is removed (in this case, blue), there is a startlingly apparent drop in image quality.
UV-B and Vitamin D3
Perhaps one of the most well known characteristics of UV-B is its role in the production of vitamin D3 and its subsequent regulation of calcium metabolism, which is needed for the development and maintenance of healthy bones (and the prevention of Metabolic Bone Disease, or MBD).
Between different species, there is wide variation between the need for dietary intake of vitamin D3 and the ability to synthesise the hormone. Vitamin D is essential for the growth and general health of many animals. In many reptile species, a vitamin D deficiency leads to the progression of MBD, which majorly impacts these animals welfare. Research on species-specific strategies for D3 acquisition has led to better advice regarding the use of UV lights with reptiles, but more research is still required as the occurrence of MBD is still common amongst certain species. It should be considered that the basking behaviours of reptiles have not only evolved to aid in thermoregulation, but as the sun is simultaneously both a heat source and a source of UV-B, basking behaviours have also evolved to help animals modulate proper exposure for sufficient production of vitamin D3. This is also true of reptiles with limited sun exposure, such as many crepuscular and nocturnal species.
Between different species, there is wide variation between the need for dietary intake of vitamin D3 and the ability to synthesise the hormone. Vitamin D is essential for the growth and general health of many animals. In many reptile species, a vitamin D deficiency leads to the progression of MBD, which majorly impacts these animals welfare. Research on species-specific strategies for D3 acquisition has led to better advice regarding the use of UV lights with reptiles, but more research is still required as the occurrence of MBD is still common amongst certain species. It should be considered that the basking behaviours of reptiles have not only evolved to aid in thermoregulation, but as the sun is simultaneously both a heat source and a source of UV-B, basking behaviours have also evolved to help animals modulate proper exposure for sufficient production of vitamin D3. This is also true of reptiles with limited sun exposure, such as many crepuscular and nocturnal species.
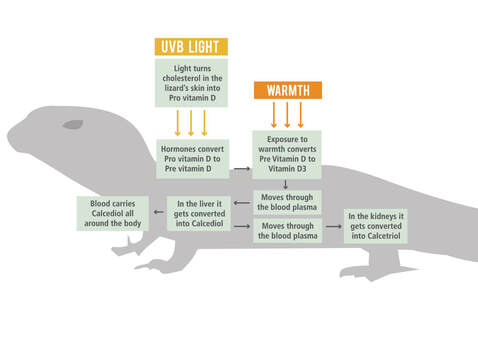
D3 Synthesis Via UV-B
Image sourced from Pinterest.com: The D3 Cycle : Arcadia Reptile
Vitamin D can be produced in the skin after exposure to UV Lighting. The precursor of vitamin D is known as pro-vitamin D3 or 7-dehydrocholesterol (7- DHC) and begins its synthesis in the malphigian layer of the skin, initiated by UV-B into pre-vitamin D3. Vitamin D3 synthesis in the skin almost all occurs between 290 – 315nm, it does not absorb ultraviolet radiation above 315 nm. Thus, sunlight with energies between 290 and 315 nm are responsible for the photolysis of pro-vitamin D3 to pre-vitamin D3. Other wavelengths may, however, have some regulatory effects on vitamin D production. This then undergoes a temperature dependant isomerisation into a stable form of vitamin D3. This process requires the skin to be at an appropriate temperature, requiring UV and heat sources to be positioned close together within a captive environment.
Regardless of whether vitamin D was sourced from the skin or digestive tract, it remains biologically inactive until its conversion by two biochemical alterations. Once in the bloodstream, the vitamin is bound to Vitamin D-Binding Proteins (VDBP) and transported to the liver where it undergoes its first hydroxylation to form calcidiol (25(OH)D). Secondary hydroxylation takes place in the kidneys to form its most biologically active metabolite, calcitriol (1,25(OH)2D).
Regardless of whether vitamin D was sourced from the skin or digestive tract, it remains biologically inactive until its conversion by two biochemical alterations. Once in the bloodstream, the vitamin is bound to Vitamin D-Binding Proteins (VDBP) and transported to the liver where it undergoes its first hydroxylation to form calcidiol (25(OH)D). Secondary hydroxylation takes place in the kidneys to form its most biologically active metabolite, calcitriol (1,25(OH)2D).
UV-B and Crepuscular Species
Historically, most studies examining the source and function of vitamin D3 have involved mammals and birds. Studies performed in reptiles have largely been focused on dietary and basking requirements in various diurnal (daytime) lizard species, but traditionally lacking attention in nocturnal (night-time)/ crepuscular (twilight), and non-lizard species. It is often assumed amongst reptile keepers that only diurnal species require exposure to UV-B. However, comparative studies between nocturnal and diurnal species demonstrate that this assumption is inaccurate.
The photobiosynthetic opportunity of the Mediterranean House Gecko (Hemidactylus turcicus) a naturally crepuscular/nocturnal species, was compared with a naturally diurnal species, the Texas Spiny Lizard (Sceloporus olivaceus) (Carmen, et al., 2000). The Spiny Lizard, being a diurnal species has a greater opportunity for exposure to UV-B but the conversion rate of pro-vitamin D3 into pre-vitamin D3 was faster in the gecko. This was early evidence that nocturnal species may have more sensitive mechanism for photobiosynthesis of D3 due to their limited exposure time. This was thought to be facilitated through the evolution of thinner skin allowing greater levels of UV-B to be absorbed.
The photobiosynthetic opportunity of the Mediterranean House Gecko (Hemidactylus turcicus) a naturally crepuscular/nocturnal species, was compared with a naturally diurnal species, the Texas Spiny Lizard (Sceloporus olivaceus) (Carmen, et al., 2000). The Spiny Lizard, being a diurnal species has a greater opportunity for exposure to UV-B but the conversion rate of pro-vitamin D3 into pre-vitamin D3 was faster in the gecko. This was early evidence that nocturnal species may have more sensitive mechanism for photobiosynthesis of D3 due to their limited exposure time. This was thought to be facilitated through the evolution of thinner skin allowing greater levels of UV-B to be absorbed.
Further investigations were carried out in a study by Ferguson, (2004) in which two species of Jamaican Anole, one shade dwelling (Anolis lineatopus merope) and the other sun dwelling (Anolis sagrei) were compared. It was once again found that the species exposed to less UV-B had a higher conversion rate of pro-vitamin D into pre-vitamin D.
|
Image taken by the author: Voluntary daytime exposure of leopard gecko under basking and UV-B light.
|
The Leopard Gecko (Eublepharis macularius), was studied directly for its ability to biosynthesise vitamin D3. Gould, et al., (2018), confirmed that Leopard Geckos exposed to short-duration (2 hour) UV-B radiation were capable of significantly increasing their vitamin D3 concentrations compared with control geckos not exposed to UV-B, but fed a similar diet. The study concludes that exposure to UV-B may help offset dietary deficiencies, or concerns about hypervitaminosis (vitamin toxicity) associated with supplements, as vitamin D3 via UV-B is a self-limiting process, and cannot result in over-supplementation, unlike oral doses of vitamin D3, as reptiles lack any regulatory mechanism for D3 uptake via the gut.
|
Additionally, skin comparison by the authors of UVGuide.co.uk revealed that the sloughed skin of the Leopard Gecko allowed in 14X more UV-B then that of the diurnal Bearded Dragon, indicating a far greater sensitivity. This is due to their limited exposure, requiring more sensitive skin to make use of what little they are exposed to. This thinner skin allows for deeper penetration of UV-B light for sufficient synthesis of vitamin D3, even under minimal lighting conditions (such as at crepuscular hours, when in shaded areas such as hiding retreats, or during brief daytime forays). This indicates a strong natural reliance on the sun to obtain adequate levels of vitamin D3.
|
In a similar study by Acierno, et al., (2008), it was found that the crepuscular Corn Snake (pantherophis guttatus, previously elaphe guttata) reacted strongly to UV-B, with D3 concentrations significantly increased in snakes that were exposed to supplemental lighting (12 hours per day). This is particularly interesting, as it had generally been assumed that basking behaviour of snakes was purely for thermoregulation, as documented by Kauffeld, (1969), although it was acknowledged in the same study that many amateur and professional keepers at the time considered UV-B to be vital for all groups of reptiles, including snakes. The assumption that snakes don't require UV-B was largely based on the fact that it was known that mammalian carnivores such as cats and dogs are unable to biosynthesise any vitamin D, so must rely totally on dietary intake. Likewise, it was assumed that because snakes are carnivores, these animals must also derive vitamin D from their diet, and that UV-B radiation–induced synthesis was likely of no importance. The results of modern studies suggest otherwise, and neglects to acknowledge just how different reptiles are to mammals.
|
Image taken by the author: corn snake basking under heat light with additional access to UV-B. - Note the presence of the log positioned on the right -hand side to facilitate 'cryptic' (partial) basking behaviour.
|
Another study by Nail, (2011) looked at whether UV-B influenced the growth rates and behaviour of hatchling Corn Snakes. The results found that those given a UV-B exposure of 2% would voluntarily expose themselves to UV-B light and that it had a positive effect on their activity levels.
Similarly, Bellamy and Stephen, (2007) found increased behavioural activity in the nocturnal Jamaican Boa (Epicrates subflavus), with the provision of UV-B lighting. Interestingly, not only are there apparent profound physiological effects after UV-B exposure, but it appears to promote psychological wellbeing as well, with the promotion of behavioural expression.
Once again these studies support the claim that species that are naturally exposed to less UV-B express the greatest rate of conversion, whilst species that are naturally exposed to the greatest degree of sunlight have the slowest rate of conversion.
It is an important consideration that reptile housing must enable the performance of an array of behaviours documented in those of free-living animals, including various basking strategies (thigmothermy and heliothermy) and the ability to regulate their own exposure to UV-B with the provision of microclimates that allow the animals to balance exposure with avoidance. However, it should not be assumed that UV-B light is a requirement for vitamin D regulation in all animals. A study in captive Royal (Ball) Pythons (Python regius) by Hedley and Eatwell, (2013) found that UV-B had no impact on D3 concentrations in that species. Although another nocturnal Python Species, the Burmese Python (Python bivittatus) was found to have a similar increase in D3 levels as that of corn snakes after exposure to UV-B, with 11 hours exposure per day (Bos, et al., 2018). These findings reaffirm that we must evaluate each species separately to determine how they acquire vitamin D so that caretakers can provide appropriate husbandry and care to improve welfare and minimise the likelihood of captive-induced diseases, as well as considerations for lighting as a source of enrichment with implications on the animals psychology, rather than just being of biological importance.
Similarly, Bellamy and Stephen, (2007) found increased behavioural activity in the nocturnal Jamaican Boa (Epicrates subflavus), with the provision of UV-B lighting. Interestingly, not only are there apparent profound physiological effects after UV-B exposure, but it appears to promote psychological wellbeing as well, with the promotion of behavioural expression.
Once again these studies support the claim that species that are naturally exposed to less UV-B express the greatest rate of conversion, whilst species that are naturally exposed to the greatest degree of sunlight have the slowest rate of conversion.
It is an important consideration that reptile housing must enable the performance of an array of behaviours documented in those of free-living animals, including various basking strategies (thigmothermy and heliothermy) and the ability to regulate their own exposure to UV-B with the provision of microclimates that allow the animals to balance exposure with avoidance. However, it should not be assumed that UV-B light is a requirement for vitamin D regulation in all animals. A study in captive Royal (Ball) Pythons (Python regius) by Hedley and Eatwell, (2013) found that UV-B had no impact on D3 concentrations in that species. Although another nocturnal Python Species, the Burmese Python (Python bivittatus) was found to have a similar increase in D3 levels as that of corn snakes after exposure to UV-B, with 11 hours exposure per day (Bos, et al., 2018). These findings reaffirm that we must evaluate each species separately to determine how they acquire vitamin D so that caretakers can provide appropriate husbandry and care to improve welfare and minimise the likelihood of captive-induced diseases, as well as considerations for lighting as a source of enrichment with implications on the animals psychology, rather than just being of biological importance.
A Further Look into the Acquisition of sufficient D3 from UV-B
Reptiles inhabit a diverse range of environmental niches, and have evolved a variety of adaptations for the acquisition of vitamin D3, either from dietary sources or UV-B. Such adaptations my be behavioural, physiological or morphological. Many species may rely entirely on UV-B to obtain sufficient levels of vitamin D3, some depend on dietary sources and others a combination. Conversely, some species may require hardly any vitamin D3 to function optimally.
Many commonly kept reptiles are from regions with high UV-B irradiance on most days of the year and would have developed mechanisms in order to take advantage of this, especially if dietary sources of D3 are scarce (such as with herbivorous or insectivorous species). Basking behaviour does not just serve as a thermoregulatory behaviour but may be modified in order to increase exposure to UV-B radiation, particularly if dietary sources are lacking. For instance, the Green Iguana (Iguana iguana) is one such species that lacks the ability to synthesise vitamin D3 in the body from dietary sources so must rely on basking behaviour. This is because its natural diet consists largely of vegetation, which, although high in vitamin D2, this metabolite is not readily bound to VDBP’s so is not usually hydroxylised into a useable form. For this reason UV-B light is absolutely required for this species to metabolise calcium properly.
The leopard gecko, an insectivorous species, is commonly provided oral supplementation of D3 as it appears adequate to meet requirements, or at least for the prevention of MBD, yet its effectiveness to meet optimal needs has not been assessed, nor has a specific dosage been investigated. Although it works, it is by no means a natural method of D3 acquisition for this species. Leopard geckos (like many crepuscular/nocturnal species) instead exhibit cathemeral behaviours, and are frequently observed active and awake during the day, at least for brief periods. This behaviour allows them brief exposure to sunlight in the wild, whether directly or in shaded areas, they have also been observed basking during the daytime in captivity. It may thusly be more appropriate to predominantly provide vitamin D3 via UV-B rather than pure supplementation.
Interestingly, a study on adult female bearded dragons revealed that weekly oral vitamin D supplementation, even at high dosages, was ineffective in attaining plasma calcidiol and calcitriol concentrations similar to UV-B exposed bearded dragons (Oonincx, et al., 2010). Yet those exposed to adequate UV-B were able to maintain stable concentrations of plasma calcidiol and calcitriol over an 83 day period after cessation of UV-B exposure, even when dietary vitamin D was low. The study states that it seems likely that during periods without UV-B exposure (e.g. lack of sunlight or ineffective artificial lamps), captive bearded dragons rely on stored reserves of vitamin D instead. This has also been observed by Acierno, et al., (2006, 2008) in similar studies on red-eared sliders and corn snakes, and may also be true for other species.
Intensity of UV-B lighting can also play a key role in the adequate acquisition of vitamin D3, for example, a study by Ferguson, et al., (2002)2 found that reproductive success of female Panther Chameleons (Furcifer pardalis) was influenced by the intensity and time exposed to UV-B lighting with the lowest rate of success, evident in those that were exposed to low levels of UV-B over a long period of time (23 hours) and those most successful were exposed to the greatest intensity of UV-B over a much shorter period of time (1 hour), indicating that light intensity is also a crucial consideration for some species, not just light duration. Baines, et al., (2016) has provided a guide known as the "UV Tool" to provide suitable recommendations of UV lighting for various reptile and amphibian species. This guide assigns various species into one of four distinct UV-B 'Zones' (Ferguson zones) based on UV-index measurements and natural UV-B exposure of the species.
As discussed already, some species conversely require only low levels of exposure as they have adapted to limited exposure with the evolution of thin, UV permeable skin. Some species may only be exposed during brief daytime forays, through cryptic basking, or limited exposure in shaded areas or dappled light. This is especially true of nocturnal and crepuscular species. It is therefore important to understand the natural basking behaviours of the species in your care in order to gauge appropriate provision of UV-B lighting and appropriate set-up to creating proper microclimates to replicate proper cover.
Despite improved welfare advice for many species, deficiencies and toxicities still occur within captive animals. It is important to note that a range of species-specific strategies are used to obtain adequate amounts of D3 for calcium homeostasis, and that this can be just as true for nocturnal and crepuscular species as it is for diurnal species.
Many commonly kept reptiles are from regions with high UV-B irradiance on most days of the year and would have developed mechanisms in order to take advantage of this, especially if dietary sources of D3 are scarce (such as with herbivorous or insectivorous species). Basking behaviour does not just serve as a thermoregulatory behaviour but may be modified in order to increase exposure to UV-B radiation, particularly if dietary sources are lacking. For instance, the Green Iguana (Iguana iguana) is one such species that lacks the ability to synthesise vitamin D3 in the body from dietary sources so must rely on basking behaviour. This is because its natural diet consists largely of vegetation, which, although high in vitamin D2, this metabolite is not readily bound to VDBP’s so is not usually hydroxylised into a useable form. For this reason UV-B light is absolutely required for this species to metabolise calcium properly.
The leopard gecko, an insectivorous species, is commonly provided oral supplementation of D3 as it appears adequate to meet requirements, or at least for the prevention of MBD, yet its effectiveness to meet optimal needs has not been assessed, nor has a specific dosage been investigated. Although it works, it is by no means a natural method of D3 acquisition for this species. Leopard geckos (like many crepuscular/nocturnal species) instead exhibit cathemeral behaviours, and are frequently observed active and awake during the day, at least for brief periods. This behaviour allows them brief exposure to sunlight in the wild, whether directly or in shaded areas, they have also been observed basking during the daytime in captivity. It may thusly be more appropriate to predominantly provide vitamin D3 via UV-B rather than pure supplementation.
Interestingly, a study on adult female bearded dragons revealed that weekly oral vitamin D supplementation, even at high dosages, was ineffective in attaining plasma calcidiol and calcitriol concentrations similar to UV-B exposed bearded dragons (Oonincx, et al., 2010). Yet those exposed to adequate UV-B were able to maintain stable concentrations of plasma calcidiol and calcitriol over an 83 day period after cessation of UV-B exposure, even when dietary vitamin D was low. The study states that it seems likely that during periods without UV-B exposure (e.g. lack of sunlight or ineffective artificial lamps), captive bearded dragons rely on stored reserves of vitamin D instead. This has also been observed by Acierno, et al., (2006, 2008) in similar studies on red-eared sliders and corn snakes, and may also be true for other species.
Intensity of UV-B lighting can also play a key role in the adequate acquisition of vitamin D3, for example, a study by Ferguson, et al., (2002)2 found that reproductive success of female Panther Chameleons (Furcifer pardalis) was influenced by the intensity and time exposed to UV-B lighting with the lowest rate of success, evident in those that were exposed to low levels of UV-B over a long period of time (23 hours) and those most successful were exposed to the greatest intensity of UV-B over a much shorter period of time (1 hour), indicating that light intensity is also a crucial consideration for some species, not just light duration. Baines, et al., (2016) has provided a guide known as the "UV Tool" to provide suitable recommendations of UV lighting for various reptile and amphibian species. This guide assigns various species into one of four distinct UV-B 'Zones' (Ferguson zones) based on UV-index measurements and natural UV-B exposure of the species.
As discussed already, some species conversely require only low levels of exposure as they have adapted to limited exposure with the evolution of thin, UV permeable skin. Some species may only be exposed during brief daytime forays, through cryptic basking, or limited exposure in shaded areas or dappled light. This is especially true of nocturnal and crepuscular species. It is therefore important to understand the natural basking behaviours of the species in your care in order to gauge appropriate provision of UV-B lighting and appropriate set-up to creating proper microclimates to replicate proper cover.
Despite improved welfare advice for many species, deficiencies and toxicities still occur within captive animals. It is important to note that a range of species-specific strategies are used to obtain adequate amounts of D3 for calcium homeostasis, and that this can be just as true for nocturnal and crepuscular species as it is for diurnal species.
References
Acierno, M., Mitchell, M., Roundtree, M., and Zachariah, T., (2006). Effects of Ultraviolet Radiation on 25-hydroxyvitamin D3 Synthesis in Red-eared Slider Turtles (Trachemys scripta elegans). American Journal of Veterinary Research, 67, 2046–2049.
Acierno, M., Mitchell, M., Zachariah, T., Roundtree, M., Kirchgessner, M. and Guzman, D., (2008). Effects of Ultraviolet Radiation on Plasma 25- hydroxyvitamin D3 Concentrations in Corn Snakes (Elaphe guttata). American Journal of Veterinary Research, 69(2), 294-297.
Ackerman, L., (1998). The Biology, Husbandry and Health Care of Reptiles (Vol I, II & III). T.F.H. Publications.
Baines, F., Chattell, J., Dale, J., Garrick, D., Gill, I., Goetz, M., Skelton, T., and Swatman, M., (2016). How much UV-B does my reptile need? The UV-Tool, a guide to the selection of UV lighting for reptiles and amphibians in captivity. Journal of Zoo and Aquarium Research, 4(1).
Bos, J., Klip, F., and Oonincx, D., (2018). Artificial Ultraviolet B Radiation Raises Plasma 25-Hydroxyvitamin D3 Concentrations in Burmese Pythons (Python bivittatus). Journal of Zoo and Wildlife Medicine, 49(3):810-812.
Chang, C., and Zheng, R., (2003). Effects of Ultraviolet B on Epidermal Morphology, Shedding, Lipid Peroxide, and Antioxidant Enzymes in Cope's Rat Snake (Elaphe taeniura). Journal of Photochemistry and Photobiology. B, Biology, 5;72(1-3):79-85.
Kauffeld, C. (1969). The Effect of Altitude, Ultra-violet Light, and Humidity on Captive Reptiles. International Zoo Yearbook. 9: 8-9
Bellamy, T., & Stephen, I., (2007). The Effect of Ultra-Violet B (UVB) Illumination and Vitamin D3 on the Activity, Behaviour and Growth Rate of the Juvenile Jamaican Boa Epicrates subflavus (Unpublished master's dissertation). University of London, United Kingdom.
Bernard, J., (1997). Vitamin D and Ultraviolet Radiation: Meeting Lighting Needs for Captive Animals. Nutritional Advisory Group Handbook.
Brames, H., (2007). Aspects of Light and Reptilian Immunity. Iguana, 14 (1), 19-23.
Carman, E., Ferguson, G., Gehrmann, G., Chen, T., and Holick, M., (2000). Photobiosynthetic Opportunity and Ability for UVB Generated Vitamin D Synthesis in Free-living House Geckos (Hemidactylus turcicus) and Texas Spiny Lizards (Sceloporus olivaceous). Copeia, 2000 (1), 245-250.
de Loos, B., (2016). Production of Cutaneous Vitamin D3 After UVB Radiation in Reptiles and Amphibians. (Unpublished dissertation).
Ferguson G., Brinker A., Gehrmann W., Bucklin S., Baines F., and Mackin S., (2010). Voluntary Exposure of Some Western-hemisphere Snake and Lizard Species to Ultraviolet-B Radiation in the Field: How Much Ultraviolet-B Should a Lizard or Snake Receive in Captivity? Zoo Biology 29: 317–334.
1Ferguson, G., Gehrmann, W., Hammack, S., Chen, T., and Holick, M., (2002). Effects of Dietary Vitamin D and UVB Irradiance on Voluntary Exposure to Ultraviolet Light, Growth and Survival of the Panther Chameleon Furcifer paradalis. Biological Effects of Light, pp193-203
2Ferguson, G., Gehrmann, W., Chen, T., Dierenfeld, E., and Holick, M., (2002). Effects of Artificial Ultraviolet Light Exposure on Reproductive Success of the Female Panther Chameleon (Furcifer pardalis) in Captivity. Zoo Biology, 21: 525-537
Ferguson, G., Gehrmann, W., Karsten, K., Hammack, S., McRae, M., Chen, T., Lung, N., and Holick, M., (2003). Do Panther Chameleons Bask to Regulate Endogenous Vitamin D3 Production? Physiological and Biochemical Zoology, 76 (1), 52-59.
Ferguson, G., Gehrmann, W., Karsten, K., Landwer, A., Carman, E., Chen, T., and Holick, M., (2004). Ultraviolet Exposure and Vitamin D Synthesis in a Sun-Dwelling and a Shade-Dwelling Species of Anolis: Are There Adaptations for Lower Ultraviolet B and Dietary Vitamin D3 Availability in the Shade? Physiological and Biochemical Zoology 78 (2).
Fleishman, L.,Loew, E., and Leal, M., (1993). Ultraviolet Vision in Lizards. Nature, 365, 397.
Girling, S., and Raiti, P., (2004). BSAVA Manual of Reptiles (second edition). BSAVA British Small Animal Veterinary Association
Gould, A., Molitor, L., Rockwell, K., Watson, M., and Mitchell, M., ( 2018). Evaluating the Physiologic Effects of Short Duration Ultraviolet B Radiation Exposure in Leopard Geckos (Eublepharis macularius). The Journal of Herpetological Medicine and Surgery, 28(1):34-39
Hedley, J., and Eatwell, K., (2013). The Effects of UV Light on Calcium Metabolism in Ball Pythons (Python regius). Veterinary Record, 173(14):345–350.
Nail, A., (2011). Does Exposure to UVB Light Influence the Growth Rates and Behaviour of Hatchling Corn Snakes, Pantherophis guttatus? (Unpublished master's dissertation). Reaseheath College and University of Chester, United Kingdom.
Nijboer, J., Van Brug, H., Tryfonidou, M., and Van Leeuwen, J., (2003). UV-B and Vitamin D3 Metabolism in Juvenile Komodo Dragons (Varanus komodoensis). Zoo Animal Nutrition, 2: 233–246.
Oishi, T., Nagai, K., Harada, Y., Naruse, M., Ohtani, M., Kawano, E., and Tamotsu, S., (2004). Circadian Rhythms in Amphibians and Reptiles: Ecological Implications. Biological Rhythm Research, 35 (1-2):105-120
Oonincx, D., Stevens, Y., van den Borne, J., van Leeuwen, J., and Hendriks, W., (2010). Effects of Vitamin D3 Supplementation and UV-B Exposure on the Growth and Plasma Concentration of Vitamin D3 Metabolites in Juvenile Bearded Dragons (Pogona vitticeps). Comparative Biochemical Physiology, Part B Biochemical Molecular Biology, 156, 122–128.
Reiter, R., Tan, D., Fuentes-Broto, L., (2010). Melatonin: A Multitasking Molecule. Progress in Brain Research, 181: 127-151
Sherwood, L., Klandorf, H., and Yancey, P., (2005). Animal Physiology: From Genes to Organisms. Thompson Brooks/Cole
Tosini, G., Bertolucci, C., and Foà, A., (2001). The Circadian System of Reptiles: A Multioscillatory and Multiphotoreceptive System. Physiology & Behaviour, 72 (2001) 461-471
Tosini, G., Ferguson, I., and Tsubota, K, (2016). Effects of Blue Light on the Circadian System and Eye Physiology. Molecular Vision: Biology and Genetics in Vision Research
Acierno, M., Mitchell, M., Zachariah, T., Roundtree, M., Kirchgessner, M. and Guzman, D., (2008). Effects of Ultraviolet Radiation on Plasma 25- hydroxyvitamin D3 Concentrations in Corn Snakes (Elaphe guttata). American Journal of Veterinary Research, 69(2), 294-297.
Ackerman, L., (1998). The Biology, Husbandry and Health Care of Reptiles (Vol I, II & III). T.F.H. Publications.
Baines, F., Chattell, J., Dale, J., Garrick, D., Gill, I., Goetz, M., Skelton, T., and Swatman, M., (2016). How much UV-B does my reptile need? The UV-Tool, a guide to the selection of UV lighting for reptiles and amphibians in captivity. Journal of Zoo and Aquarium Research, 4(1).
Bos, J., Klip, F., and Oonincx, D., (2018). Artificial Ultraviolet B Radiation Raises Plasma 25-Hydroxyvitamin D3 Concentrations in Burmese Pythons (Python bivittatus). Journal of Zoo and Wildlife Medicine, 49(3):810-812.
Chang, C., and Zheng, R., (2003). Effects of Ultraviolet B on Epidermal Morphology, Shedding, Lipid Peroxide, and Antioxidant Enzymes in Cope's Rat Snake (Elaphe taeniura). Journal of Photochemistry and Photobiology. B, Biology, 5;72(1-3):79-85.
Kauffeld, C. (1969). The Effect of Altitude, Ultra-violet Light, and Humidity on Captive Reptiles. International Zoo Yearbook. 9: 8-9
Bellamy, T., & Stephen, I., (2007). The Effect of Ultra-Violet B (UVB) Illumination and Vitamin D3 on the Activity, Behaviour and Growth Rate of the Juvenile Jamaican Boa Epicrates subflavus (Unpublished master's dissertation). University of London, United Kingdom.
Bernard, J., (1997). Vitamin D and Ultraviolet Radiation: Meeting Lighting Needs for Captive Animals. Nutritional Advisory Group Handbook.
Brames, H., (2007). Aspects of Light and Reptilian Immunity. Iguana, 14 (1), 19-23.
Carman, E., Ferguson, G., Gehrmann, G., Chen, T., and Holick, M., (2000). Photobiosynthetic Opportunity and Ability for UVB Generated Vitamin D Synthesis in Free-living House Geckos (Hemidactylus turcicus) and Texas Spiny Lizards (Sceloporus olivaceous). Copeia, 2000 (1), 245-250.
de Loos, B., (2016). Production of Cutaneous Vitamin D3 After UVB Radiation in Reptiles and Amphibians. (Unpublished dissertation).
Ferguson G., Brinker A., Gehrmann W., Bucklin S., Baines F., and Mackin S., (2010). Voluntary Exposure of Some Western-hemisphere Snake and Lizard Species to Ultraviolet-B Radiation in the Field: How Much Ultraviolet-B Should a Lizard or Snake Receive in Captivity? Zoo Biology 29: 317–334.
1Ferguson, G., Gehrmann, W., Hammack, S., Chen, T., and Holick, M., (2002). Effects of Dietary Vitamin D and UVB Irradiance on Voluntary Exposure to Ultraviolet Light, Growth and Survival of the Panther Chameleon Furcifer paradalis. Biological Effects of Light, pp193-203
2Ferguson, G., Gehrmann, W., Chen, T., Dierenfeld, E., and Holick, M., (2002). Effects of Artificial Ultraviolet Light Exposure on Reproductive Success of the Female Panther Chameleon (Furcifer pardalis) in Captivity. Zoo Biology, 21: 525-537
Ferguson, G., Gehrmann, W., Karsten, K., Hammack, S., McRae, M., Chen, T., Lung, N., and Holick, M., (2003). Do Panther Chameleons Bask to Regulate Endogenous Vitamin D3 Production? Physiological and Biochemical Zoology, 76 (1), 52-59.
Ferguson, G., Gehrmann, W., Karsten, K., Landwer, A., Carman, E., Chen, T., and Holick, M., (2004). Ultraviolet Exposure and Vitamin D Synthesis in a Sun-Dwelling and a Shade-Dwelling Species of Anolis: Are There Adaptations for Lower Ultraviolet B and Dietary Vitamin D3 Availability in the Shade? Physiological and Biochemical Zoology 78 (2).
Fleishman, L.,Loew, E., and Leal, M., (1993). Ultraviolet Vision in Lizards. Nature, 365, 397.
Girling, S., and Raiti, P., (2004). BSAVA Manual of Reptiles (second edition). BSAVA British Small Animal Veterinary Association
Gould, A., Molitor, L., Rockwell, K., Watson, M., and Mitchell, M., ( 2018). Evaluating the Physiologic Effects of Short Duration Ultraviolet B Radiation Exposure in Leopard Geckos (Eublepharis macularius). The Journal of Herpetological Medicine and Surgery, 28(1):34-39
Hedley, J., and Eatwell, K., (2013). The Effects of UV Light on Calcium Metabolism in Ball Pythons (Python regius). Veterinary Record, 173(14):345–350.
Nail, A., (2011). Does Exposure to UVB Light Influence the Growth Rates and Behaviour of Hatchling Corn Snakes, Pantherophis guttatus? (Unpublished master's dissertation). Reaseheath College and University of Chester, United Kingdom.
Nijboer, J., Van Brug, H., Tryfonidou, M., and Van Leeuwen, J., (2003). UV-B and Vitamin D3 Metabolism in Juvenile Komodo Dragons (Varanus komodoensis). Zoo Animal Nutrition, 2: 233–246.
Oishi, T., Nagai, K., Harada, Y., Naruse, M., Ohtani, M., Kawano, E., and Tamotsu, S., (2004). Circadian Rhythms in Amphibians and Reptiles: Ecological Implications. Biological Rhythm Research, 35 (1-2):105-120
Oonincx, D., Stevens, Y., van den Borne, J., van Leeuwen, J., and Hendriks, W., (2010). Effects of Vitamin D3 Supplementation and UV-B Exposure on the Growth and Plasma Concentration of Vitamin D3 Metabolites in Juvenile Bearded Dragons (Pogona vitticeps). Comparative Biochemical Physiology, Part B Biochemical Molecular Biology, 156, 122–128.
Reiter, R., Tan, D., Fuentes-Broto, L., (2010). Melatonin: A Multitasking Molecule. Progress in Brain Research, 181: 127-151
Sherwood, L., Klandorf, H., and Yancey, P., (2005). Animal Physiology: From Genes to Organisms. Thompson Brooks/Cole
Tosini, G., Bertolucci, C., and Foà, A., (2001). The Circadian System of Reptiles: A Multioscillatory and Multiphotoreceptive System. Physiology & Behaviour, 72 (2001) 461-471
Tosini, G., Ferguson, I., and Tsubota, K, (2016). Effects of Blue Light on the Circadian System and Eye Physiology. Molecular Vision: Biology and Genetics in Vision Research